![]()
Prevention is better than Treatment
OSMMR2000-D G-6-PD KIT
Take a clean 96-well (preferably U bottom) microplate (elution microplate).
Add 5 microliters of whole blood or one disk cut from a dried blood spot (3/16 inch or 2 X 1/8inch diameter) per well. You can use one microplate for all kind of samples (whole blood and dried blood spots can be measured simultaneously - see note on step 4 below). Remember to add controls (not included in this kit)*
Add 75 microliters of Elution Buffer in each well, mix well the contents of each well and place the plate in a plate shaker (preferably in an incubator at 37 degrees C). Let the Color reagent warm up to room temperature.
Wait 20 minutes for dried blood spots or 8 minutes for whole blood samples. Hint : if performing an analysis of both dried blood spots and whole blood samples in the same microplate, you can start the elution of the dried blood spots and add the elution buffer in the whole blood samples 8 minutes before the elution stage is over.
While waiting reconstitute one Reagent Vial with Distilled Water. Add 40 ml / Reagent vial if you are using the 4X500 kit format or 8 ml if you are using the 20X100 kit format.
Add 75 microliters of Reagent to each well in a new microplate (reagent microplate).
Slowly warm the microplate with the reagents up to the temperature you will run the experiment (preferably in an incubator at 37 degrees C)
Once the elution stage is over, take both microplates out of the incubator and transfer 15 microliters of each eluted sample to the corresponding well of the reagent microplate (already containing the reagents). Mix well the contents of each well.
Immediately take one single reading at 405 nm (Hb evaluation).
Place the reagent microplate in the incubator and wait for 5-10 minutes.
Add 100 microliters of Color Reagent per well. Mix well.
Place the microplate in the microplate reader. The reader should be set to "endpoint" mode (single measurement) at 570 nm (550-570nm). Take two readings at time = 0 and time = 10 minutes. In the meantime, the microplate should remain in the incubator. Calculate the total ΔOD for each sample and the controls. If your reader is equipped with a kinetic program and a heating device, you can measure the plate in kinetic mode at 550-570 nm. If you use the kit designated " 492 nm " then the reading should be taken at 492 nm. Temperature fluctuations may have a significant effect on the readings so, unless a heating device is present, the endpoint protocol is encouraged.
Use the formulas at the end of this page to express your results directly in U / g Hb. (Greek Patent # 1003227; International Patent Pending)(upper: 2 point mode, bottom: kinetic mode)
Notes
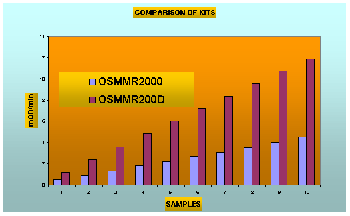
on this chart:
The chart above shows the results
obtained from samples with progressively higher G-6-PD activity
using the two different kits, the OSMMR2000 (340 nm - kinetic mode)
and OSMMR2000D (550 nm - two point measurement - endpoint mode).
Temperature was set to 37 C and the light path was 1 cm. In a
microplate reader the mOD/min obtained were reduced by 50% for
both kits (light path approximately 4 mm). Data
produced by R&D Diagnostics Ltd.
Endpoint (two-point) mode:
(δODsample550nm)/(δODcontrol550nm)
----------------------------------------------------------
X Control=Sample (U/g Hb)
ODsample405nm / ODcontrol405nm
Kinetic mode
(δODsample550nm/min)/(δODcontrol550nm/min)
----------------------------------------------------------
X Control=Sample (U/g Hb)
ODsample405nm / ODcontrol405nm
INSTRUCTIONS FOR USE (PACKAGE INSERT)
R&D DIAGNOSTICS LTD ®

A SCREENING KIT FOR DRIED BLOOD SPOTS
This kit is particularly suitable for screening for G-6-pd deficiency in newborns.
WITH Hb NORMALIZATION© PROCEDURE

MANUFACTURER CODE: GRM7 - EDMA CODE: 13.01.09.01 - PRODUCT FILE No. GR/CA01/GRM7/O/127
KIT SIZES: 2000 / 75 TESTS
Manufactured by :
R & D DIAGNOSTICS LTD
33, Alevizatou street,
15669 Papagos
Greece
Version XIX, 5 NOVEMBER 2004
Catalog No : OS-MMR2000-D (One Step kit 550 nm)
BY
R&D Diagnostics Limited, Papagos, Greece.
www.rddiagnostics.com
CONTENTS
1) Intended Use
2) Clinical Background (Summary and Explanation of the Test)
3) Principle of the Assay
4) Kit Contents
5) Reagents / Materials required but not provided
6) Precautions - Safety and Operating
7) Sample Collection
8) Reagent Reconstitution
9) Assay Procedures
10) Calculation of Results
11) Quality Assurance
12) Expected Values
13) Comparative Protocol Evaluation
14) Performance Characteristics
15) Limitations of Use
16) Summary of Protocols
17) References
1. Intended Use
The OSMMR-D-D assay is an enzymatic colorimetric method for the quantitative determination of G-6-pd activity in dried blood spots specimens. The test is intended for use as a screening method for red cell glucose-6-phosphate dehydrogenase deficiency in newborns.
2. Clinical Background (Summary and Explanation of the Test)
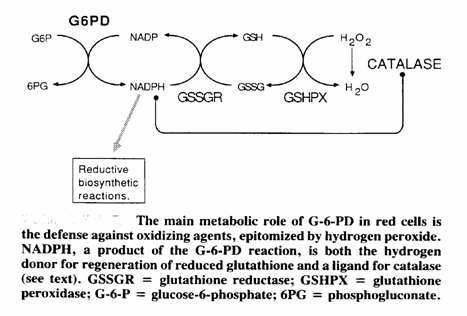
Glucose-6-phosphate dehydrogenase (G-6-PD) is a cytoplasmic enzyme that is distributed in all cells. It catalyses the first step in the hexose monophosphate pathway, producing NADPH (Picture 1). This coenzyme is required as hydrogen donor for reactions of various biochemical pathways as well as for the stability of catalase and the preservation and regeneration of the reduced form of glutathione. Catalase and glutathione are both essential for the detoxification of hydrogen peroxide, therefor the defence of cells against H2O2 ultimately and heavily depends on G-6-PD. The red cells are exquisitely sensitive for oxidative damage and lack other NADPH-producing enzymes. The defence against oxidizing agents, epitomised by H2O2, is mainly realised by glutathione, which converts H2O2 to H2O stoichiometrically via glutathione peroxidase. NADPH is the hydrogen donor for the regeneration of reduced glutathione. An alternative pathway of H2O2 detoxification is via catalase, but this route is regarded ineffective under normal conditions (1), because of the lower affinity of catalase for H2O2 compared to that of glutathione peroxidase. G-6-PD deficiency is the most common known enzymopathy with around 400 million people affected worldwide. The prevalence ranges from 5 to 25% in endemic areas, such as Africa (2,3), the Midle East, Asia (4,5,6,7), the Mediterranean (8,9,10) and Papua New Guinea. The highest incidence is found in Kurdish Jews: 65%. Incidences ranging from 0.5 to 6.9% have been reported in North and South America. Around 400 mutations have been reported so far (11,12,13,14).
The clinical manifestations associated with G-6-PD deficiency are:
Drug induced hemolysis: certain antimalarials, sulfonamides, sulfones and other drugs or chemicals are associated with significant hemolysis in subjects (15,16,17).
Infection induced hemolysis: numerous bacterial, viral and rickettsial infections have precipitated hemolysis, but the most important are infectious hepatitis, pneumonia and typhoid fever (18,19,20).
Favism: sudden onset of acute hemolytic anemia within 24 to 48 hours of ingesting fava beans (21,22, 23, 24, 25, 26, 28).
Neonatal jaundice: jaundice usually appears by 1 to 4 days of age (29,30,31,32).
Chronic nonspherocytic hemolytic anemia (33).
3. Principle of the Assay
The OSMMR-D G-6-PD Assay utilizes glucose-6-phosphate-dehydrogenase, which in the presence of NADP, catalyses the oxidation of glucose-6-phosphate to 6-phosphogluconate. The NADPH produced reacts with a color reagent in which a tetrazolium salt gets reduced producing a distinct color. This color is measured colorimetrically at 550 nm (500-570 nm) and is directly proportional to the concentration of Glucose-6-phosphate dehydrogenase present in the sample. The results are calculated by evaluating the increase in OD per minute (slope) for unknowns against the slope for a Standard with known G-6-PD activity. Alternatively, the reduced tetrazolium salt can be measured in endpoint mode using two measurements one at time = 0 and a second one at time = 10-15 minutes later. During the measurements the microplate should remain in the incubator. This protocol is strongly encouraged if your microplate reader is not equipped with a kinetic software or a heating plate. In this case the total ΔOD / sample should be used for calculations. See the attached "Benchtop" instructions for the formulas.
4. Kit Contents
The OSMMR-D G-6-pd kit contains rubber stoppered glass vials secured with an aluminum crimp seal, which are labeled accordingly. In the large kits (2000 tests) a transparent plastic bottle with a screw cap is included. The whole kit is better stored refrigerated. The kit has enough reagents for at least 2000 or 75 tests. The kit comes in three formats: 4 X 50ml reagent vials (each sufficient for at least 500 tests); 20 X 8ml reagent vials (each sufficient for at least 100 tests) or 3 X 2 ml vials (each sufficient for at least 25 tests).
Kit for 2000 tests
4 X 40 ml ELUTION BUFFER (Code RD6001), Liquid. Contains 0.15% (w/v) sodium azide as preservative. (CAUTION: See safety section 6). Store at room temperature. Refrigeration recommended..
4 X 40 ml or 20 X 8 ml REAGENT VIALS LYOPHILIZED (Code RD6002). Store at 2-8o C (refrigerated – do not freeze) CAUTION: Very toxic to humans.
1 X 17.5 ml COLOR REAGENT BOOSTER vial (liquid, Code RDC0301). Store at 2-8o C (refrigerated – do not freeze)
1 X 175 ml COLOR REAGENT bottle (transparent, bright yellow liquid; Code RDC8003). Store at 2-8o C (refrigerated – do not freeze) CAUTION: Keep away from strong light sources.
Kit for 75 tests
1 X 6 ml ELUTION BUFFER (Code RD6001), Liquid. Contains 0.15% (w/v) sodium azide as preservative. (CAUTION: See safety section 6). Store at room temperature. Refrigeration recommended..
3 X 2 ml REAGENT VIALS LYOPHILIZED (Code RD6002). Store at 2-8o C (refrigerated – do not freeze) CAUTION: Very toxic to humans.
1 X 1 ml COLOR REAGENT BOOSTER vial (liquid, Code RDC0301). Store at 2-8o C (refrigerated – do not freeze)
1 X 7 ml COLOR REAGENT bottle (transparent, bright yellow liquid; Code RDC8003). Store at 2-8o C (refrigerated – do not freeze) CAUTION: Keep away from strong light sources.
Stability of the Chemicals
The minimum shelf-life of the chemicals contained in the kit are as follows :
ELUTION BUFFER Stable for at least 24 months at room temperature.
See Exp.date on sticker.
REAGENT VIAL Stable for 5 days (after reconstitution) at 2-8oC, stable for 5 days
frozen (after reconstitution) plus an additional 5 days following
thawing. Stable for at least 18 months lyophilized in unopened vial. See Exp.date on sticker.
COLOR REAGENT BOOSTER Stable for at least 24 months refrigerated. See Exp.date on
sticker.
COLOR REAGENT Stable for at least 24 months refrigerated. See Exp.date on
sticker.
Although some of the chemicals are stable at room temperature, storing at 2-8oC is highly recommended for all chemicals. This will reduce degradation even more and slow down any microbial infections.
Ongoing stability studies may prove that the chemicals can be stored for much longer periods under certain conditions. This information will be included in the updated temporary insert leaflets and in the final version.
5. Reagents / Materials required but not provided
1. U - well micro titration plates (elution plates)
2. Water for injection (de-ionized water).
3. Flat-bottomed micro titration plates (assay plates)
4. Single or multichannel automatic pipettes to deliver volumes in the range of 15 to 75μl with an accuracy of +/- 1.5% over this range.
5. A micro titration plate reader capable of reading absorbances at 340 nm in KINETIC reading mode.
6. A hole punch which produces 3/16” or 1/8” diameter discs.
7. A plate incubator set at 37oC with an accuracy of +/- 1oC
8. Blood collection cards. The minimum preprinted information required (in accordance to Schleicher and Schuell) is :
1. Infant’s name, Mother’s name and Patient ID number
2. Date of Birth
3. Birth Weight
4. Sex
5. Feeding status
6. Specimen collection date
7. Submitter’s ID and address
8. Physician’s name and telephone number
9. Testing laboratory, name and address
9. Space for test results
10. Appropriate number of preprinted ½” (internal diameter) broken or dotted line circles on attached blood collection filter paper.
11. Manufacturer’s and lot number of filter paper indicated on filter paper
12. Manufacturer or printer listed on the information section.
Blood collection filter cards should be attached to at least 2 copies of the information section outlined above.
Filter paper should can be used according to the specification provided by the National Screening Association.
6. Precautions - Safety and Operating
All reagents must be stored according to the instructions on the label and instructions for use.
The assay-performance can be seriously affected if the instructions as outlined in this package insert and on the labels are not strictly adhered to.
The Elution Agent contains 0.15% sodium azide as anti microbial preservative. This substance has a toxic effect if absorbed or indigested. They should be discarded with an adequate water flow.
All blood samples of human origin must be regarded as a potential biohazard s and normal laboratory precautions must be taken whilst handling these samples.
Do not use kit components after the expiry date stated on the label.
Do not mix components of different lot numbers
Do not use any solutions that have become turbid or discolored.
7. Sample Collection
Collect from the infant's heel according to the NCCLS Approved Standard LA4-A2 "Blood Collection on Filter Paper for neonatal Screening Programs" Second edition (1992) NCCLS, Villanova, PA
After the sample is taken and the blood has dried, the cards must be stored at 2-8oC. Spots not stored under these conditions gradually loose the enzyme activity due to heat inactivation, causing potential risk of misclassifying samples as screen-positive (34,35).
8. Reagent Preparation
The reagents must be warmed up slowly to 30 or 37oC prior to use if these are the temperatures the assay is going to be performed at. No pre-warming needed for temperatures 22-29oC.
Reconstitute 1 VIAL of REAGENT with 40 ml of WATER FOR INJECTION (or de-ionized water). If you are using the 20X100 test format, each reagent vial should be reconstituted with 8 ml of WATER FOR INJECTION (or de-ionized water). Use 2 ml if you are using the 25 test / vial kits. The number of tests / vial is shown on the label attached on the reagent vial.
Stable for 5 days at 2-80C or 10 days at -200C. If a frozen aliquot is thawed, this is stable for 4 days at 2-80C
Prepare a working mixture of color reagent. To do this mix 1 part of Color Reagent Booster with 10 parts of Color Reagent. Return the original components in the refrigerator. The Color Reagent Mixture prepared this way is stable for at least 8 hours.
9. Assay Procedure
1) Punch blood spots 3/16 inch diameter (or 2x 1/8" ) in U bottom micro titer plate. Alternatively, you can use 5 microliters of whole blood (with anticoagulant).
Use position A1, A2 for the Normal Standard, (not provided with the kit).
Optionally, you may use 2 wells for the Intermediate and Deficient Controls (not provided with the kit.)
2) Add 75 μl of ELUTION agent to each well.
3) Place micro titer plate on an orbital plate shaker for 30 minutes at room temperature (8 minutes for whole blood samples). This step should be performed at Room Temperature.
4) During the elution, reconstitute and prepare Reagent Mixture. The reagents must be warmed up slowly to 30 or 37oC prior to use if this is the temperature the assay is going to be performed at.
5) Transfer 15μl of the eluant from each well to the corresponding well of a new flat bottom microtiter plate. If out of range values are obtained in step 10 below, the transfer volume can be reduced to 10 microliters.
6) Add 75 μl of the pre-warmed reagent to each well and mix well.
7) Add 80 μl the Color Reagent Mixture prepared by mixing 1 part of Color Reagent Booster with 10 parts of Color Reagent.
8) Read the plate in a plate reader at 550 nm (500-570 nm) for 12-15 minutes with 60 seconds intervals. The reader must be fitted with a temperature-controlled incubator. Alternatively, the NADPH produced can be measured in endpoint mode using two measurements one at time = 0 and a second one at time = 12-15 minutes later. During the measurements the microplate should remain in the incubator. This protocol is strongly encouraged if your microplate reader is not equipped with a kinetic software or a heating plate. In this case the total OD / sample should be used for calculations. See the attached "Benchtop" instructions for the formulas.
10) After the readings are completed, change the program of the reader to an ENDPOINT MODE select wavelength = 405 nm and read the plate (containing the same mixture) once again. See section 10 “OBTAINING RESULTS DIRECTLY EXPRESSED IN U/g Hb”
10. Calculation of Results
The optical densities for the 12-15 measurements per sample are plotted against time (in minutes) from which the increase in OD (slopes) for each well can be calculated in OD/min.
These slopes are evaluated against the slope for a Standard with known G-6-PD activity (mean of the slopes of the duplicates). The value for the Standard is divided by slope for the Standard to obtain a factor. This factor is used to multiply the slope's obtained for the unknowns:.
factor = value Control / slope Control
slope unknown x factor = U G-6-PD activity
Example: mean slope Normal Control = 0.015829 OD/min.
value Control = 17.0 U/mgHb
factor = 17.0 / 0.015829 = 1074
slope sample = 0.00845 G-6-PD activity = 9.075 U
OBTAINING RESULTS DIRECTLY EXPRESSED IN U / g Hb. (37, 38, 39)
The following formula expresses your results directly into U / g Hb (Greek Patent # 1003227; International Patent Pending)
(dOdsample550nm/min)/(dOdcontorl550nm/min)
![]() x Control
Value = Sample Value (Activity in U/g Hb)
x Control
Value = Sample Value (Activity in U/g Hb)
Odsample405nm/ODcontrol405nm
wherein dODsample is the change in optical density (per minute) for the sample, 550 nm is the wavelength at which optical density is measured under kinetic mode, dODcontrol is the change in optical density (per minute) of a control measured under kinetic mode conditions at a particular wavelength (550 nm), ODsample is the optical density for the sample measured once at a particular wavelength (405nm), and ODcontrol is the optical density of the control measured once at that particular wavelength (405nm), 405nm is the wavelength at which a reading of Heamoglobin contained can be taken, the control value being determined by the change in optical density (per minute) of the control measured under kinetic mode at 550nm divided by the optical density of the control at 405nm i.e. dODcontrol55nm/mim/ODcontrol405nm.
The Kit described above was used to demonstrate the effectiveness of the second reading on a Cobas Mira Plus ( Roche Diagnostics ). The experiment was set up to mimic the effect of incomplete elution on the measurement of enzyme activity. According to the assay protocol described above 2 spots of 3.2mm must be punched from the sample and inserted into the sample cup. This was done using whole blood samples of 3 human donors spotted and dried onto filtration paper. The instrument was programmed for the second reading by repeating the assay from the same sample, only with a single reading at 405nm instead of the kinetic reading at 550 nm. To mimic the effect of incomplete elution the same samples were used but only one spot was punched and inserted into the sample cup.
The results are shown below in Table 1:
Table 1
|
|
|
mOD/min 550nm |
|
|
|
405nm |
|
|
|
|
Units |
|
2 spot |
1 spot |
% |
|
1 spot |
2 spot |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
MMRC02 |
N |
19.42 |
9.98 |
51.4% |
|
1.81 |
3.28 |
55.3% |
|
|
MMRC02 |
N |
23.18 |
11.2 |
48.3% |
|
1.92 |
3.81 |
50.5% |
|
|
USA |
1 |
8.29 |
4.2 |
50.7% |
|
1.04 |
2.07 |
50.2% |
|
|
USA |
1 |
7.76 |
4.71 |
60.7% |
|
1.12 |
1.97 |
56.8% |
|
|
USA |
2 |
12.98 |
5.98 |
46.1% |
|
1.16 |
2.48 |
46.9% |
|
|
USA |
2 |
13.05 |
6.07 |
46.5% |
|
1.14 |
2.40 |
47.7% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ratio |
|
|
|
|
Normalised |
|
|
|
|
|
2 spot |
1 spot |
|
|
1 spot |
2 spot |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
MMRC02 |
N |
1.000 |
1.810 |
|
|
18.06 |
19.42 |
93.0% |
|
|
MMRC02 |
N |
1.162 |
1.705 |
|
|
19.10 |
19.96 |
95.7% |
|
|
USA |
1 |
0.631 |
3.154 |
|
|
13.25 |
13.14 |
100.8% |
|
|
USA |
1 |
0.601 |
2.932 |
|
|
13.81 |
12.92 |
106.9% |
|
|
USA |
2 |
0.756 |
2.823 |
|
|
16.88 |
17.17 |
98.3% |
|
|
USA |
2 |
0.732 |
2.866 |
|
|
17.40 |
17.84 |
97.6% |
|
11. Quality Assurance
This kit has been thoroughly checked before shipment. Moreover all kits are checked at regular intervals during their shelf life to ensure compliance to the specifications set. The reproducibility of the control values should be within defined limits of laboratory acceptability. Commonly used measures of variability are discussed by Westgard, J.O., et al.36 If the precision of the assay does not correlate with this standard and repetition excludes errors in technique, check the following areas:
a. Pipetting and timing devices
b. Instrument calibration
c. Expiration dates on reagent labels and prepared working solutions
d. Storage conditions
e. Temperature control devices
f. Wrong use (or no use) of the Hemoglobin normalization procedure.
12. Expected Values
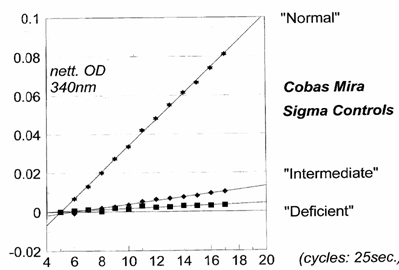
Values Obtained
|
Cycle No. |
Deficient * |
Intermediate * |
Normal * |
|
5 |
0 |
0 |
0 |
|
6 |
0.0060 |
-0.0008 |
0.0067 |
|
7 |
0.0011 |
0.0009 |
0.0130 |
|
8 |
0.0001 |
0.0019 |
0.0200 |
|
9 |
0.0019 |
0.0025 |
0.0273 |
|
10 |
0.0015 |
0.0035 |
0.0335 |
|
11 |
0.0029 |
0.0052 |
0.0420 |
|
12 |
0.0022 |
0.0062 |
0.0481 |
|
13 |
0.0024 |
0.0068 |
0.0551 |
|
14 |
0.0030 |
0.0075 |
0.0615 |
|
15 |
0.0029 |
0.0081 |
0.0667 |
|
16 |
0.0030 |
0.0094 |
0.0740 |
|
17 |
0.0033 |
0.0102 |
0.0812 |
· Values are expressed in ΔODs
13. Comparative Protocol Evaluation
This kit was compared against the Sigma kit and showed an excellent correlation of the results obtained. Results are given in the graph below. Please note that the results of the Sigma are expressed in U / L while results with our kit are in U / g Hb.
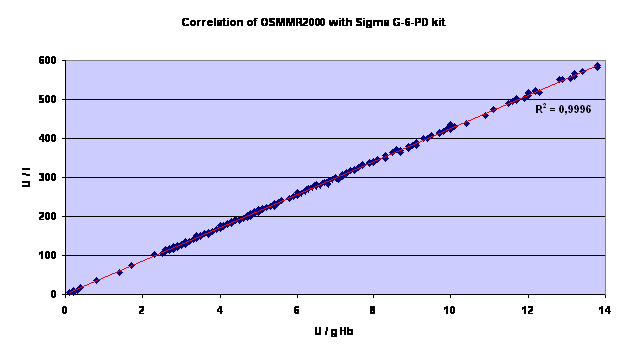
14. Performance Characteristics
See attached Quality Control Certificate issued for this batch.
15. Limitations of Use
Low results are not diagnostic per se of G-6-pd deficiency but indicate the need for further study of the newborn from which a presumptive screen positive sample was received.
Important note: Do not perform the assay at temperatures over 37oC as this may inactivate the enzyme and lead to erroneous results.
In case of bubble formation in the well a FALSE NEGATIVE result may occur, caused by diffusion of the light passing vertically through the well leading to falsely high absorption of light. As a consequence, a G-6-PD Deficient specimen could be misclassified as NORMAL.
In some forms of G-6-PDH deficiency, young erythrocytes manifest normal enzyme activity. Blood from patients who have just experienced a hemolytic crisis must first be treated by the procedure described by Herz et al (37) to separate the older erythrocytes from the prevailing population of young ones. Use 0,005 ml of the suspension so obtained for the assay.
If the patient has received a blood transfusion, this test is clinically significant only after 30 days have elapsed, because the donor's erythrocytes generally manifest a normal G-6-PDH activity and can thus bias the result before the expiration of this time.
16. Summary of Protocol
SEE ATTACHED BRIEF INSERT
17. References
References in the Text
1.Cohen G. and Hochstein P. Generation of hydrogen peroxide in erythrocytes by haemolytic agents. Biochemistry, 3:895, 1964.
2. Capps F.P.A., Gilles H.M., Jolly H. and Worlledge S.M. Glucose-6-phosphate dehydrogenase deficiency and neonatal jaundice in Nigeria. Their relation to the prophylactic use of vitamin K. Lancet, ii:379, 1963.
3. Bienzle U., Effiong C.E. and Luzzatto L. Erythrocyte glucose-6-phosphate dehydrogenase deficiency (G6PD type A-) and neonatal jaundice. Acta Paed Scand 65:701, 1976.
4. Lai H.C. Lai M.P.Y. and Leung K.S. Glucose-6-phosphate dehydrogenase deificiency in Chinese. J Clin Pathol 21:44, 1968.
5. Lu T-C, Wei H. and Blackwell R.Q. Increased incidence of severe hyperbilinurinaemia among newborn Chinese infants with G6PD deficiency. Pediatrics 37:994, 1966
6. Flatz G., Sringam S., Premyothin C., Penbharkkul S., Ketusingh R. and Chulajata R. Glucose 6-phosphate dehydrogenase deficiency and neonatal jaundice. Arch Dis Child 38:566, 1963.
7. Phornphutkul C., Whitaker J.A. and Woramthumrong N. Severe hyperbilinurinemia in Thai newborns in association with erythrocyte G6PD deficiency. Clin Pediatr 8:275, 1969.
8. Doxiades S.A. and Valaes F. The clinical picture of Glucose 6-phosphate dehydrogenase deficiency in early infancy. Arch Dis Child 39:545, 1964.
9. Doxiades S.A., Fessas P.H. and Valaes F. Erythrocyte enzyme deficiency in unexplained kernicterus. Lancet ii:44, 1960.
10.Meloni T., Cagnazzo G., Dore A. and Cutillo S. Phenobarbital for prevention of hyperbilinurinemia in Glucose 6-phosphate dehydrogenase deficient newborn infants. J Pediatr 82:1048, 1973.
11.Matthay K.K and Mentzer W.C. Erythrocyte enzymopathies in the newborn. Clin Haematol 10:31, 1981.
12.Malluh A.A., Imseeh G., Abu-Osba Y.K.and Hamdan J.A. Screening for Glucose 6-phosphate dehydrogenase deficiency can prevent severe neonatal jaundice. Ann Trop Paed 12:391, 1992.
13.Solem E. Glucose 6-phosphate dehydrogenase deficiency : An easy and sensitive quantitative assay for the detection of female heterozygotes in red blood cells. Clin Chim Acta 142:153, 1984.
14.Solem E., Pirzer C., Siege M., Kollman F., Romero - Savaria O., Barktsch - Trefs O. and Kornhuber B. Mass screening for Glucose 6-phosphate dehydrogenase deficiency. Improved fluorescent spot test. Clin Chim Acta 152:135, 1985.
15.Dern R.J., Beutler E. and Alving A.S. The haemolytic affect of primaquine. II. The natural course of the haemolytic anaemia and the mechanisms of its self-limiting character. J Lab Clin Med 44:171, 1954.
16.Beutler E., Dern R.J. and Alving A.S. The haemolytic effect of primaquine. IV. The relationship of cell age to hemolysis. J Lab Clin Med 44:439, 1954.
17.Beutler E. The hemolytic effect of primaquine and related compounds. Blood, 14:103, 1959.
18.Chan T.K., Chesterman C.N., McFadzean A.J.S. and Todd D. The survival of Glucose 6-phosphate dehydrogenase deficient erythrocytes in patients with typhoid fever on chloramphenicol therapy. J Lab Clin Med 77:177, 1971.
19.Burka E.R., Weaver Z. and Marks P.A. Clinical spectrum of hemolytic anemia associated with Glucose 6-phosphate dehydrogenase deficiency. Ann Intern Med 64:817, 1966.
20.Owusu S.K., Addy J., Foli A.K., Janosi M., Konotey - Ahulu F.I.D. and Larbi E.B. Acute reversible renal failure associated with Glucose 6-phosphate dehydrogenase deficiency. Lancet 1:1255, 1972
21. Fermi C. and Martinetti P. Studio sul favismo. Ann.Igiene Sper, 15:76, 1905
22.Luisada L. Favism: A singular disease affecting chiefly red blood cells. Medicine, 20:229, 1941
23.Sansone G., Piga A.M. and Segni G. Il Favismo, Torino, Italy, Minerva Medica, 1958.
24.Kattamis C.A., Kyriazakou M. and Chaidas S. Favism : Clinical and biochemical data. J Med Genet 6:34, 1969.
25.Kahn A., Marie J., Desbois J.C. and Boivin P. Favism in a Portuguese family due to a deficient Glucose 6-phosphate dehydrogenase variant of Canton or Canton-like type 1. Acta Haematol 56:58, 1976.
26. Belsey M.A. The epidemiology of favism. Bull WHO 48:1, 1973.
27.Szeinberg A., Sheba C., Hirschorn N. and Bodonyi E. Studies on erythrocytes in cases with past history of favism and drug induced avute hemolytic anemia. Blood 12:603, 1957.
28.Gross A.T., Hurwitz R.A. and Marks P.A. An hereditary enzymatic defect in erythrocyte metabolism. Glucose 6-phosphate dehydrogenase deficiency. J Clin Invest 37:1176, 1958.
29.Matthay K.K. and Mentzer W.C. Erythrocyte enzymopathies in the newborn. Clin Haematol 10:31, 1981.
30.Smith G.D. and Vella F. Erythrocyte enzyme deficiency in unexplained kernicterus. Lancet 1:1133, 1960.
31.Bienzle U. Glucose 6-phosphate dehydrogenase deficiency. Part I : Tropical Africa. Clin Haematol 10:785, 1981.
32.Karayalcin C., Acs H. and Lanzkowsky P. G6PD deficiency and hyperbilinurinemia in black American full-term infants. NY State J Med 79:22, 1979.
33.Luzzatto L. Inherited haemolytic states : Glucose 6-phosphate dehydrogenase deficiency. Clin Hematol 4:83, 1975.
34.Motulsky A.G., Campbell - Kraut J.M. Population genetics of Glucose 6-phosphate dehydrogenase deficiency of the red cell in : Blumberg B.S. (ed): Proceedings of Conference on Genetic Polymorphisms and Geographic Variations in Disease. New York: Grune & Stratton, 1961, p.159.
35.Bernstein R.E. Brillinat cresyl blue screening test for demonstrating Glucose 6-phosphate dehydrogenase deficiency in red cells. Clin Chim Acta 8:158, 1963.
36. Westgard, J.O., et al. 1981 Clin. Chem. 27:493-501.
37. Herz F, Kaplan E, Scheye ES. Diagnosis of erythrocyte glucose-6-phosphate dehydrogenase deficiency in the negro male despite hemolytic crisis. Blood 1970;35:953-954.
38. Reclos G.J., Hatzidakis C.J. and Kruithof R.A. G-6-PD Diagnosis : Modification of the standard method eliminates the need for an additional Hemoglobin determination. Pharmakeftiki 12(1): 25-31, 1999
39. Reclos G.J., Hatzidakis C.J. and Schulpis K.H. Glucose-6-phosphate dehydrogenase deficiency neonatal screening: preliminary evidence that a high percentage of partially deficient female neonates are missed during routine screening. J Med Screening, 7(1): 46-51, 2000
40. Reclos G.J., Schulpis K.H., Gevrili S. and Vlachos G. Evaluation of glucose-6-phosphate dehydrogenase activity in two different ethnic groups using a kit employing the haemoglobin normalization procedure. Clin Biochemistry, 36: 393-5, 2003.
General References
[1] Beutler E. Study of Glucose-6-Phosphate dehydrogenase : History and Molecular Biology. Am.J.Hematol., 42:53, 1993.
[2] Lucio Luzzatto and Atui Mehta, “Glucose 6-Phosphate Dehydrogenase Deficiency, pp. 3367-3398, in The Metabolic and Molecular Bases of Inherited Disease, by Scriver C.R. et al. McGraw and Hill Editions, 1994.
Contact Information
For Technical Details, Article reprints, Lists of Contraindicated Drugs and other Assay Information you may contact us by
Mail: Send to R&D DIAGNOSTICS LTD, 33, ALEVIZATOU STREET, 15669 PAPAGOS, GREECE.
Phone: Dial +30-210-6537307 or +30-210-6547434
Fax: Send at +30-210-6548284
E-mail info@rddiagnostics.com
You may find more information on this Disease and a step by step example of the correct use of the Normalization Procedure in our site at
About us The Disorders The Products Kit Inserts Literature Technical Support Articles Events Links Contact us To Order
Hosted by
ohmywebsite.com
TO
SEARCH THIS SITE - CLICK
SEARCH
Page last edited on 21/03/2006